Calcium Carbonate Characteristics
Compound Name: Calcium Carbonate
Molecular Formula: CaCO3
Mass: 100.0869 g/mol
Density: 2.71g/cm3 (calcite)
2.83g/cm3
Melting point: 825°C (aragonite)
1339°C (calcite)
Boiling Point: decomposes
Solubility in water: 0.00015mol/L (25°C)
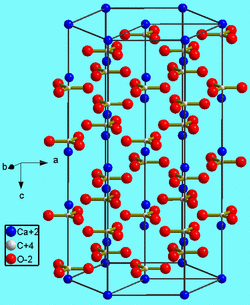
Structure:
Molecular Formula: CaCO3
Mass: 100.0869 g/mol
Density: 2.71g/cm3 (calcite)
2.83g/cm3
Melting point: 825°C (aragonite)
1339°C (calcite)
Boiling Point: decomposes
Solubility in water: 0.00015mol/L (25°C)
Structure:
- Crystal structure: Trigonometrical
- Percent Composition by mass: 64%
- Bond: covalent
- reacts strongly with acids, and reactions form carbon dioxide.
- releases carbon dioxide when heated to above 840°C and forms calcium oxide
- forms soluble calcium bicarbonate when it reacts with water that is saturated with carbon dioxide.
- carbon bicarbonate - Ca(HCO3)2 - is essential to the erosion of carbonate rock
